Chapter 2
Chemistry of Life
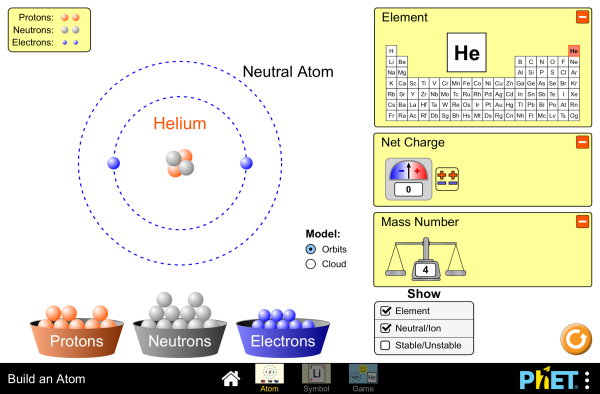
Everything is made of tiny particles called atoms; from the bones in your body to the air that you breathe, it's all made of atoms and groups of atoms put together like Legos. You should learn the parts of the atoms, how they combine together, and which ones are most common to living creatures. An ordered chart of all the known elements is called the periodic table. P-table is another very good resource.
Build some atoms.
The number of electrons in an element affect it's bonding capabilities and potential energy levels. These are modeled in orbits around the nucleus for the sake of understanding them but each orbit in truth is a representation of a likely location for those electrons and not an assigned static position. Atoms join together through covalent and ionic bonding and create other substances with unique physical and chemical properties.
Lets practice our Ionic bonding skills.
Everything is made of tiny particles called atoms; from the bones in your body to the air that you breathe, it's all made of atoms and groups of atoms put together like Legos. You should learn the parts of the atoms, how they combine together, and which ones are most common to living creatures. An ordered chart of all the known elements is called the periodic table. P-table is another very good resource.
Build some atoms.
The number of electrons in an element affect it's bonding capabilities and potential energy levels. These are modeled in orbits around the nucleus for the sake of understanding them but each orbit in truth is a representation of a likely location for those electrons and not an assigned static position. Atoms join together through covalent and ionic bonding and create other substances with unique physical and chemical properties.
Lets practice our Ionic bonding skills.
| |||||||
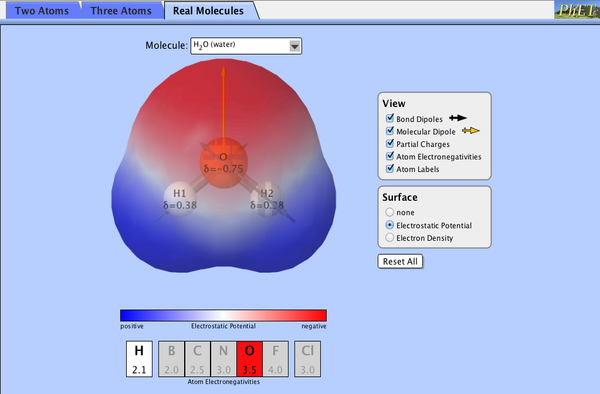
Water
They say water is the stuff of life. Well what makes it so special? Water has certain properties that make it important in solutions of all kinds. At it's heart is the wondrous hydrogen bond.
They say water is the stuff of life. Well what makes it so special? Water has certain properties that make it important in solutions of all kinds. At it's heart is the wondrous hydrogen bond.
Carbon and all of its bonds
Carbon is versatile due to it's many bonding sites in its outer electron ring. Those multiple combinations allow for the more complex molecules required for life.
Carbon is versatile due to it's many bonding sites in its outer electron ring. Those multiple combinations allow for the more complex molecules required for life.
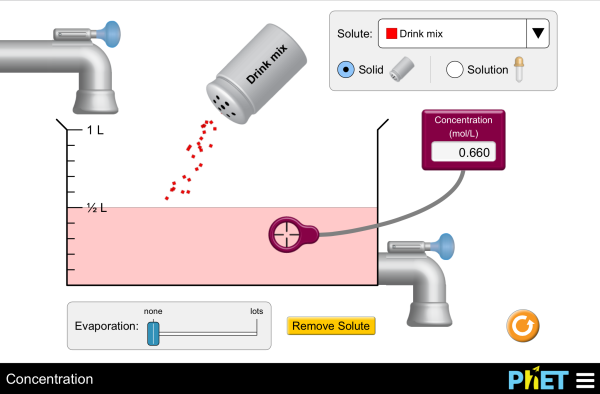
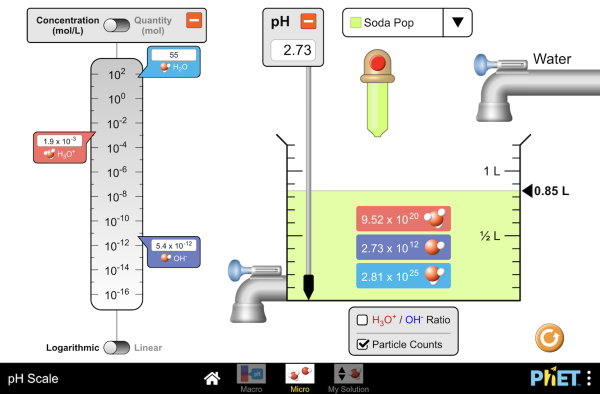
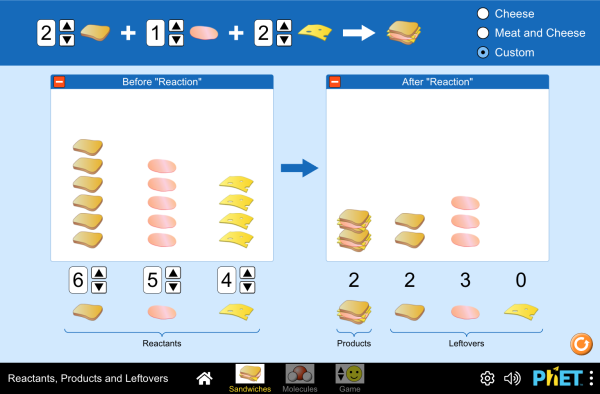
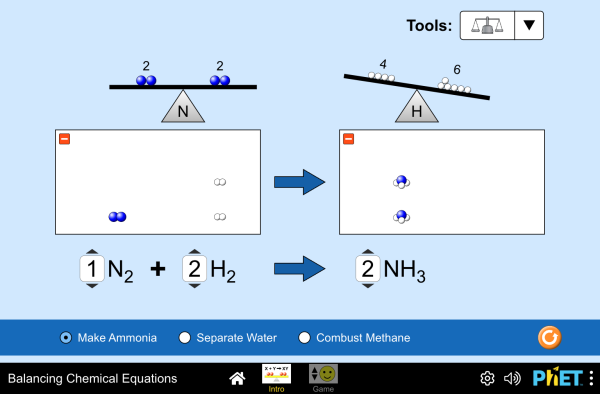
Balance is the Key to the Universe
Every equation, by definition, must be balanced. What goes in must come out- nothing more, nothing less. You can practice with the simulators below for visual presentation of the formulas.
Every equation, by definition, must be balanced. What goes in must come out- nothing more, nothing less. You can practice with the simulators below for visual presentation of the formulas.
| |||||||
Think you've got it? Here are some more places to practice.
PCLC- practice your knowledge of common compounds and balance them with visual supports.
Creative Chemistry- Fill in the proper coefficients to balance the formulas.
MPC- Yet another fill in the blank coefficient balancer.
Jefferson Lab- still more to choose from.
PCLC- practice your knowledge of common compounds and balance them with visual supports.
Creative Chemistry- Fill in the proper coefficients to balance the formulas.
MPC- Yet another fill in the blank coefficient balancer.
Jefferson Lab- still more to choose from.
| 2_macromolecule_slogans.pdf | |
| File Size: | 36 kb |
| File Type: | |
Don't forget the world of enzymes.
| enzyme_foldable_instructions.pdf | |
| File Size: | 197 kb |
| File Type: | |